With hydrogen’s widely recognised role as a safe and clean energy carrier, research into its production, particularly from renewable sources, has intensified. One of several pathways to generate hydrogen is the separation of water using sunlight with the help of photocatalysts. Partially supported by the EU-funded DYNAPORE project, researchers have synthesised a new organic material for photocatalytic solar hydrogen production, or water splitting. Their findings were published recently in the journal ‘Nature Chemistry’.
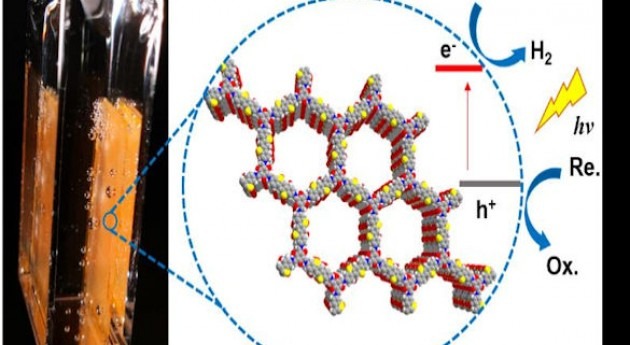
According to a news release posted on the University of Liverpool website, the team combined experiments and computation for developing the photocatalyst. Photocatalytic water splitting using sunlight is seen as a promising technology that can provide high-energy yield without pollutant by-products. This is possible only if the energy in sunlight can be harvested effectively, as explained in the news release. “Inorganic materials are better known as water splitting catalysts, but organic catalysts can also be built from cheap abundant elements, such as carbon, nitrogen, and sulphur.” Xiaoyan Wang, a chemistry PhD student who led the experimental work at the University, said: “To achieve high hydrogen evolution rates, you need good water affinity, broad light adsorption, high surface area, and high crystallinity. By introducing all of these features in one material, we got a very active photocatalyst.”
Stability and efficiency
In the journal article, the researchers noted that in nature, organic molecules are used “for light harvesting and photosynthesis, but most man-made water splitting catalysts are inorganic semiconductors.” They added: “Organic photocatalysts, while attractive because of their synthetic tunability, tend to have low quantum efficiencies for water splitting. Here we present a crystalline covalent organic framework (COF) based on a benzo-bis (benzothiophene sulfone) moiety that shows a much higher activity for photochemical hydrogen evolution than its amorphous or semicrystalline counterparts.”
They emphasised that the “COF is stable under long-term visible irradiation and shows steady photochemical hydrogen evolution with a sacrificial electron donor for at least 50 hours.” The scientists attributed the high quantum efficiency of “fused-sulfone-COF to its crystallinity, its strong visible light absorption, and its wettable, hydrophilic 3.2 nm mesopores.” They argued that these pores’ impact on the framework led to “a further 61% enhancement in the hydrogen evolution rate up to 16.3 mmol g−1 h−1. The COF also retained its photocatalytic activity when cast as a thin film onto a support.”
COFs have entered the spotlight recently as highly promising hydrogen evolution photocatalysts. These are a relatively new class of crystalline organic polymers that are made up of light elements such as oxygen, boron and nitrogen, and are connected by strong covalent bonds to form rigid, porous structures. Covalent bond refers to the interatomic linkage that results from the sharing of an electron pair between two atoms. COFs’ application potential in many fields, including catalysis, organic electronics and energy storage, has made them an attractive research topic.
The DYNAPORE (Dynamic responsive porous crystals) project that provided funding for the study aims “to develop synergic, multidisciplinary experimental and computational capability to harness the dynamics of flexible crystalline porous solids for function, demonstrated in separation and catalysis,” according to CORDIS. In addition, it “addresses the long-term vision of man-made materials with chemical selectivity and functional efficiency produced by dynamic structural flexibility.”