Refractory organic pollutants, including phenols, perfluorinated compounds, and antibiotics, are abundant in various industrial wastewater streams such as chemical, pharmaceutical, coking, and dyeing sectors, as well as municipal and domestic sources. These pollutants pose significant threats to ecological well-being and human health.
The imperative to achieve complete removal of organic contaminants from water and facilitate water recycling is paramount for enhancing environmental quality and ensuring sustainable economic and social progress. Addressing the efficient removal of recalcitrant organic pollutants in water is not only a focal point in environmental chemical pollution control research but also a pivotal technical challenge constraining industrial wastewater reuse.
Advanced oxidation processes (AOPs), especially heterogeneous AOPs, yield strongly reactive oxygen species including ·OH, ·O2-, and ·SO4- to oxidize organic pollutants under ambient conditions, are appealing wastewater treatment technologies for decentralized systems. AOPs often need excessive energy input (UV light or electricity) to activate soluble oxidants (H2O2, O3, persulfates), thus more cost-effective AOPs are urgently required.
By virtue of ease of separation and utilization of sunlight, heterogeneous photocatalysis is becoming a sustainable and promising AOP strategy for addressing environmental issues.
Widely used inorganic photocatalysts exhibit robust stability and efficient mineralization activities. However, their broad bandgap, which restricts the sunlight absorption range, and low adsorption capacity for organic pollutants collectively impair the overall efficiency of pollutant photodegradation.
In contrast, organic semiconductors represented by g-C3N4 offer the advantage of extended-spectrum utilization and excellent adsorption ability due to their high surface area and substantial π-π stacking. Nevertheless, their utilization is hampered by the generation of high binding-energy Frenkel excitons upon light excitation, impeding the separation of long-lived photogenerated electrons and hole efficiency.
The limited carrier separation efficiency significantly curtails the photodegradation activity of organic photocatalysts. The shallow valence band positions of these photocatalysts also limit their mineralization efficiency. Furthermore, compared with established wastewater treatment methods like Fenton or Fenton-like processes, photocatalytic technologies often exhibit low treatment capacity, markedly falling short of industrialization demands.
Based on the relevant scientific issues within photocatalytic wastewater treatment, recently, a research team led by Prof. Yongfa Zhu from Tsinghua University, China, summarized their progress in pollutant degradation using organic photocatalysts to propel the practical implementation of photocatalytic water treatment and served as a reference for researchers in this field.
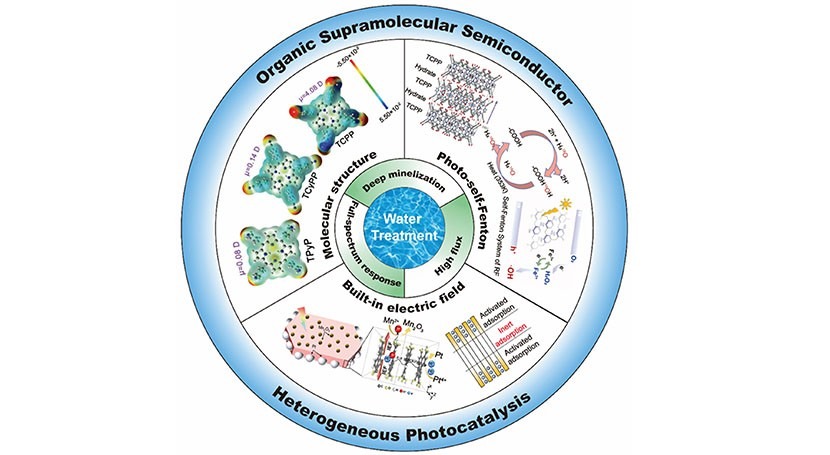
Firstly, they developed new supramolecular and polymeric organic photocatalytic systems to elevate light utilization efficiency. By modulating the effect of monomer structure on energy band position, they expanded the absorption scope to the near-infrared region, realizing mineralization under solar light.
Secondly, they unveiled the role of dipoles and crystalline order in modulating the built-in electric field, enabling efficient charge migration from bulk to surface, thus significantly enhancing pollutant degradation and mineralization rates.
Finally, they established a novel approach for photo-self-Fenton high-flux mineralization of organic pollutants to improve the treatment capacity of photodegradation and overcome the limitations of the Fenton method. The new system combines in situ H2O2 generation through photocatalytic redox reaction with in situ Fenton reaction synergistically, which achieves high-flux mineralization under visible light without additional oxidants, thereby elevating pollutant mineralization from 30% to over 90%.
The results were published in Chinese Journal of Catalysis.